A UC San Diego research team has discovered how a molecule on the outside of the SARS-Cov-2 spike protein acts as a "gate" for the virus that causes COVID-19 infection, which could help find a way to counter the virus; all that's needed is a "key" to lock it, a new study says.
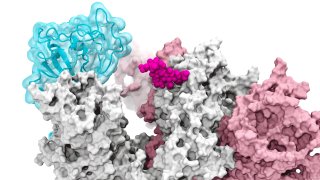
The research published Thursday in the journal "Nature Chemistry," described how glycans -- molecules that make up a sugary residue around the edges of the spike protein -- can allow the virus to enter and infect healthy human cells.
"We essentially figured out how the spike actually opens and infects," said UCSD's Rommie Amaro, a computational biophysical chemist, who helped develop a detailed visualization of the SARS-CoV-2 spike protein that efficiently latches onto our cell receptors.
"We've unlocked an important secret of the spike in how it infects cells. Without this gate, the virus basically is rendered incapable of infection," said Amaro, a professor of chemistry and biochemistry and a senior author of the new study.
Get San Diego local news, weather forecasts, sports and lifestyle stories to your inbox. Sign up for NBC San Diego newsletters.
Amaro said she believes the research team's gate discovery opens potential avenues for new therapeutics to counter SARS-CoV-2 infection. If glycan gates could be pharmacologically "locked" in the closed position, then the virus is effectively prevented from opening to entry and infection.
Others on the project include co-senior author Lillian Chong at the University of Pittsburgh, first author and UCSD graduate student Terra Sztain and co-first author and UCSD postdoctoral scholar Surl-Hee Ahn.
The spike's coating of glycans helps deceive the human immune system since it comes across as nothing more than a sugary residue, according to the research. Previous technologies that imaged these structures depicted glycans in static open or closed positions, which initially didn't draw much interest from scientists. Supercomputing simulations then allowed the researchers to develop dynamic "movies" that revealed glycan gates activating from one position to another, offering an unprecedented piece of the infection story.

"We were actually able to watch the opening and closing," said Amaro. "That's one of the really cool things these simulations give you -- the ability to see really detailed movies.
Local
"When you watch them, you realize you're seeing something that we otherwise would have ignored," she continued. "You look at just the closed structure, and then you look at the open structure, and it doesn't look like anything special. It's only because we captured the movie of the whole process that you actually see it doing its thing."
The simulations were first run on "Comet" at the San Diego Supercomputer Center at UCSD and later on "Longhorn" at University of Texas, Austin. Such computing power provided the researchers with atomic-level views of the spike protein receptor binding domain, or RBD, from more than 300 perspectives. The investigations revealed glycan "N343" as the linchpin that pries the RBD from the "down" to "up" position to allow access to the host cell's receptors. The researchers describe the glycan activation as similar to a "molecular crowbar" mechanism.



