Why the NFL Gave UC San Diego Half a Million Dollars to Study Cannabis as a Treatment for Injuries
-

CDC investigating botched Botox shots in 9 states
At least 19 women in nine states reportedly became sick after they got Botox, either having gotten the injections from people who were never licensed or trained to give the shots or received them in &... -

These foods don't deserve their bad reputations, dietitians and doctors say
Eggs, potatoes, coffee: These kitchen staples, among others, have gained bad reputations, nutrition experts say, but don’t necessarily deserve it. In fact, registered dietitians, doctors and nutrition... -

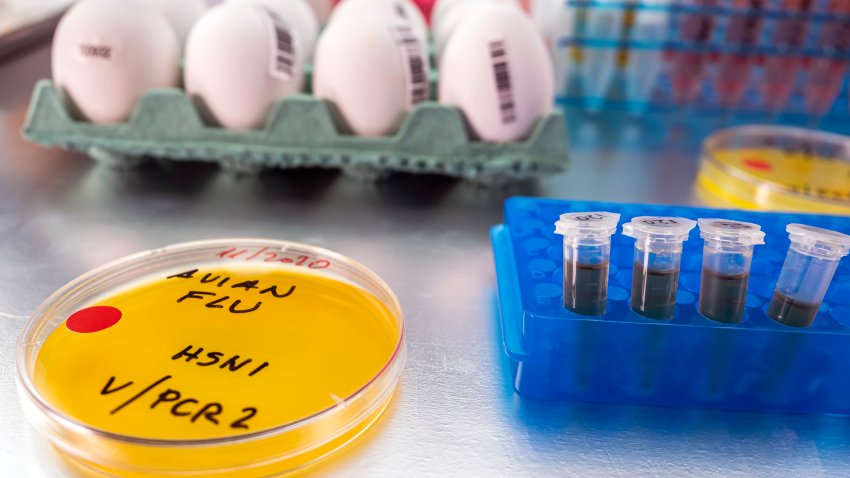
Bird flu is spreading to more farm animals. Are milk and eggs safe?
A bird flu outbreak in U.S. dairy cows has spread to more than two dozen herds in eight states. That comes weeks after the nation’s largest egg producer found the virus in its chickens. Health officia... -
Dartmouth researchers look to meld therapy apps with modern AI
Therabot, currently in its first clinical trial, uses generative AI trained on therapy scripts in an effort to create technology that brings mental health services to underserved populations.