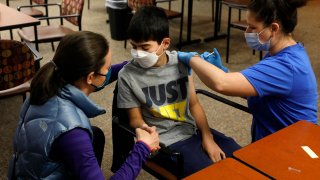
- Pfizer and BioNTech's Covid vaccine is safe and appears to generate a robust immune response in a clinical trial of kids 5 to 11, the drugmakers announced.
- The companies tested a two-dose regimen of 10 micrograms — about a third the dosage used for teens and adults — administered three weeks apart.
- They plan to submit the data to the FDA and other health regulators "as soon as possible."
A smaller dose of Pfizer and BioNTech's Covid-19 vaccine is safe and generates a "robust" immune response in a clinical trial of kids ages 5 to 11, the drugmakers announced Monday.
The news couldn't come any sooner for parents anxious to get their children vaccinated as kids start the new school year with the delta variant surging across America. Children's Covid cases remain disturbingly high with 243,000 new infections during the week ended Sept. 9. — the second-highest number of kids' cases since the beginning of the pandemic, according to the most recent data from the American Academy of Pediatrics.
Get San Diego local news, weather forecasts, sports and lifestyle stories to your inbox. Sign up for NBC San Diego newsletters.
The data, which included more than 2,200 children, will be submitted to the Food and Drug Administration and other health regulators "as soon as possible," the companies said. Pfizer CEO Albert Bourla said last week the company could submit data on children ages 5 to 11 by the end of this month. If the FDA spends as much time reviewing the data for that age group as it did for 12- to 15-year-olds, the shots could be available in time for Halloween.
"Depending on how long the FDA takes to review the application, whether it's a four-week review or a six-week review, you could have a vaccine available to children as early as probably by the end of October" or early November, Scott Gottlieb, a Pfizer board member and the former head of the FDA, told CNBC on Monday.
Money Report
The companies tested a two-dose regimen of 10 micrograms — about a third the dosage used for teens and adults — administered three weeks apart. They said the shots were well tolerated and produced an immune response and side effects comparable to those seen in a study of people ages 16 to 25.
Common side effects for teens and adults include fatigue, headache, muscle pain, chills, fever and nausea, according to the Centers for Disease Control and Prevention.
"We are eager to extend the protection afforded by the vaccine to this younger population, subject to regulatory authorization, especially as we track the spread of the Delta variant and the substantial threat it poses to children," Bourla said in a press release that announced the results.
The companies didn't disclose many details about the trial, including whether any of the kids in the trial experienced myocarditis, a rare heart condition seen in a small number of adolescents and young adults.
So far, the Pfizer-BioNTech coronavirus vaccine has been cleared by the FDA for people as young as 12, while Moderna's and Johnson & Johnson's vaccines have been authorized for adults.
The FDA is expected to issue a decision sometime this week on which groups are eligible to get a third dose, or booster shot, of the Pfizer vaccine. An FDA advisory committee on Friday unanimously recommended Pfizer booster shots to people age 65 and older and other vulnerable Americans.
Additionally, Pfizer expects to release clinical trial data on how well its Covid vaccine works in children as young as 6 months to 5 years old as early as the end of October, he said.






